We saw in the previous section that n-pentane has the most elongated structure and that this increased surface area available for contact between molecules resulted in a stronger London force. 
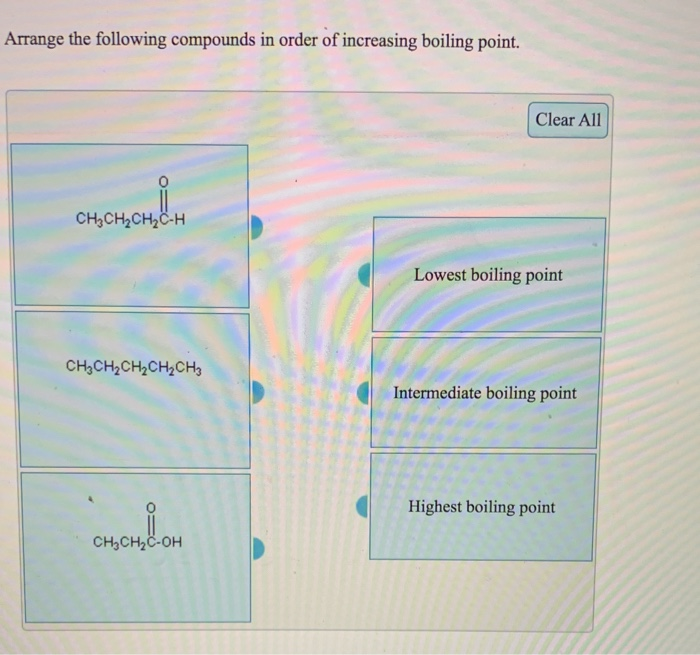
It may be useful to revisit the structures. Explain how you came to your conclusions. Using what you know about how London forces are affected by the shape of a molecule, determine which boiling point corresponds to which isomer. The boiling points of these isomers are 9.5 ☌, 27 ☌ and 36 ☌, in no particular order. The previous section compared the isomers isopentane, n-pentane, and neopentane and ranked the strength of their IMFs. The increase in melting and boiling points with increasing atomic/molecular size may be rationalized by considering how the strength of dispersion forces is affected by the electronic structure of the atoms or molecules in the substance.Įffect of Shape on London Forces and Boiling PointsĪs seen in the previous module, the shapes of molecules also affect the magnitudes of the dispersion forces between them. Melting and Boiling Points of the Halogens Halogen Examining the melting and boiling points for these halogens shows a trend that larger and heavier atoms display stronger IMFs, as shown by the higher melting and boiling points. A higher melting and boiling point indicates that more kinetic energy is required to overcome the IMFs and melt or boil the substance. But how might we compare the IMFs between F 2 and Cl 2, two gases at the same temperature and pressure? To do this, we can make use of melting point and boiling point data, as seen in Table 1. We determined that I 2 must have the strongest IMFs due to existing in the solid state and that F 2 and Cl 2 must have the weakest IMFs. All of these molecules have London forces as their primary IMF since they are nonpolar molecules. In the previous section, we compared F 2 and Cl 2 (gases at room temperature) to Br 2 (a liquid at room temperature), and I 2 (a solid at room temperature). In this section we expand on these concepts. We also used the fact that HCl(ℓ) has a higher boiling point than F 2(ℓ) as evidence that it has stronger IMFs. 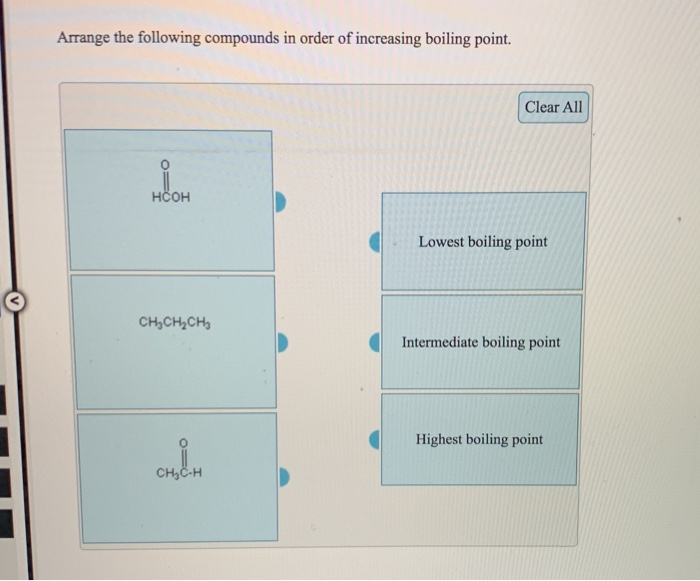
In the previous section, we discussed how we could qualitatively compare the strength of IMFs between two substances if we knew that they were different phases at the same temperature and pressure. | Key Concepts and Summary | End of Section Exercises | Melting Point and Boiling Point Comparisons | Melting Point and Boiling Point Comparisons | Effect of Hydrogen Bonding on Boiling Point | Predict relative melting or boiling points of similar compounds.Connect trends in physical properties with relative strength of intermolecular forces.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
